Applications
 Part of the Oxford Instruments Group
Part of the Oxford Instruments Group
Expand
Collapse
 Part of the Oxford Instruments Group
Part of the Oxford Instruments Group
Neuroscience
In a recent study, Dr. Charles L. Limoli and colleagues at the University of California, Irvine, used Imaris software to quantify radiation-induced changes to microvascular in the brain. The work revealed that the automation available with Imaris software can greatly expedite the imaging and analysis of microvessel parameters in 3D.
The researchers carried out the study because they were interested in quantifying how irradiation used for cancer treatment affected microvascular in central nervous system structures. The ability to quantify vascular abnormalities that result from such treatments can help assess injury and determine the success of interventions designed to protect the vasculature and surrounding microenvironment. Such interventions might lead to efficacious therapies for treatment or preventing the damage that can result from radiation and/or chemotherapy treatments.
One software package, many tools
Although the researchers in Dr. Limoli’s laboratory frequently use the Imaris FilamentTracer module to trace and and reconstruct neurons, they have also found it useful for other applications because of its many analysis and statistical quantification tools — such as Surface rendering, Colocalization, Filament analysis (XTension), and Spots detection.
“Our initial interest in Imaris was driven by its ability to image and quantify dendrites and neuron spine density,” said Erich Giedzinski, who manages the lab and conducts analyses with Imaris. “But we realized that Imaris could also assist us with numerous applications including cell counting, colocalization quantification, and spine classification, just to name a few.”
The researchers decided to compare Imaris and another software package to better understand the ability of each to reconstruct the microvasculature of an irradiated and unirradiated rodent hippocampus, a region of the brain critical for learning and memory.
For the Imaris portion of the work, the researchers imported deconvolved confocal microscopy images into Imaris. They used the Imaris Surface tool to construct a 3D model of the vasculature that helped eliminate a small amount of background and channel signal crossover that could potentially interfere with the Filament rendering. Once the Surface was constructed, they created a mask of the Surface rendering so that they could easily remove all the unwanted signal. The Surface structures were then traced with the FilamentTracer tool to determine conglomerate length, area, and volume.
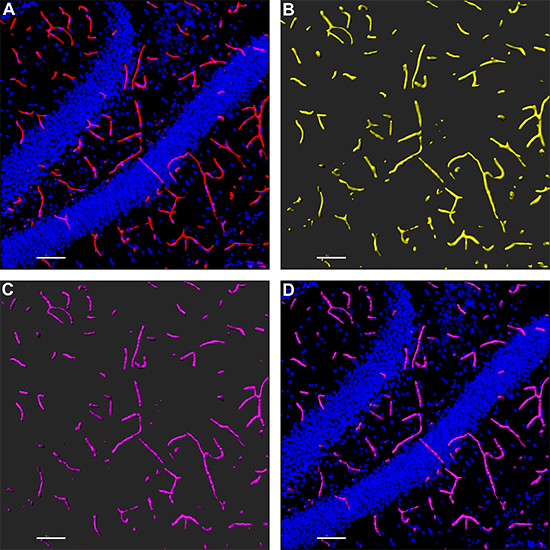
A) The researchers started with a confocal image of the dentate gyrus hippocampal region of a 6-month-old rat, with microvasculature stained with tomato lectin (red) and nuclei counterstained with DAPI (blue). B) They then used Imaris for Surface rendering of the microvasculature. C) This was followed by Filament modeling of the microvasculature from the Surface rendering. D) The Filament model is combined with the original DAPI image. Images courtesy of Erich Giedzinski, University of California, Irvine.
Short- and long-term changes in microvasculature
The quantitative image analysis revealed that irradiation caused significant short- and long-term reductions in capillary density, diameter, and volume. The researchers think that these changes may contribute, or even cause, some of the adverse neurocognitive side effects seen in cancer patients after radiotherapy treatment of a brain tumor.
While both software packages tested by the researchers provided detailed structural analyses of the microvasculature, the ability of Imaris to automate surface analysis, including 3D rendering, provided significant time savings.
“Frequent updates to the software have decreased analysis times and greatly improved the output of data in our lab,” said Giedzinski. “The technical assistance we’ve received from Bitplane in terms of utilizing new tools and new applications has also been outstanding.”
Author: Dr. Charles L. Limoli and colleagues at the University of California, Irvine
Category: Case Study
