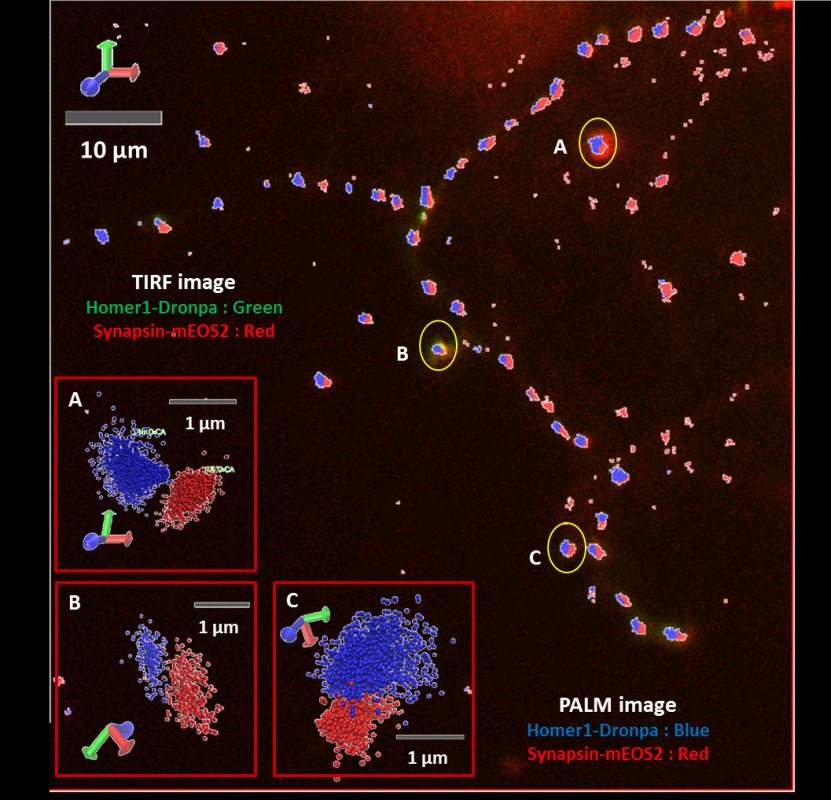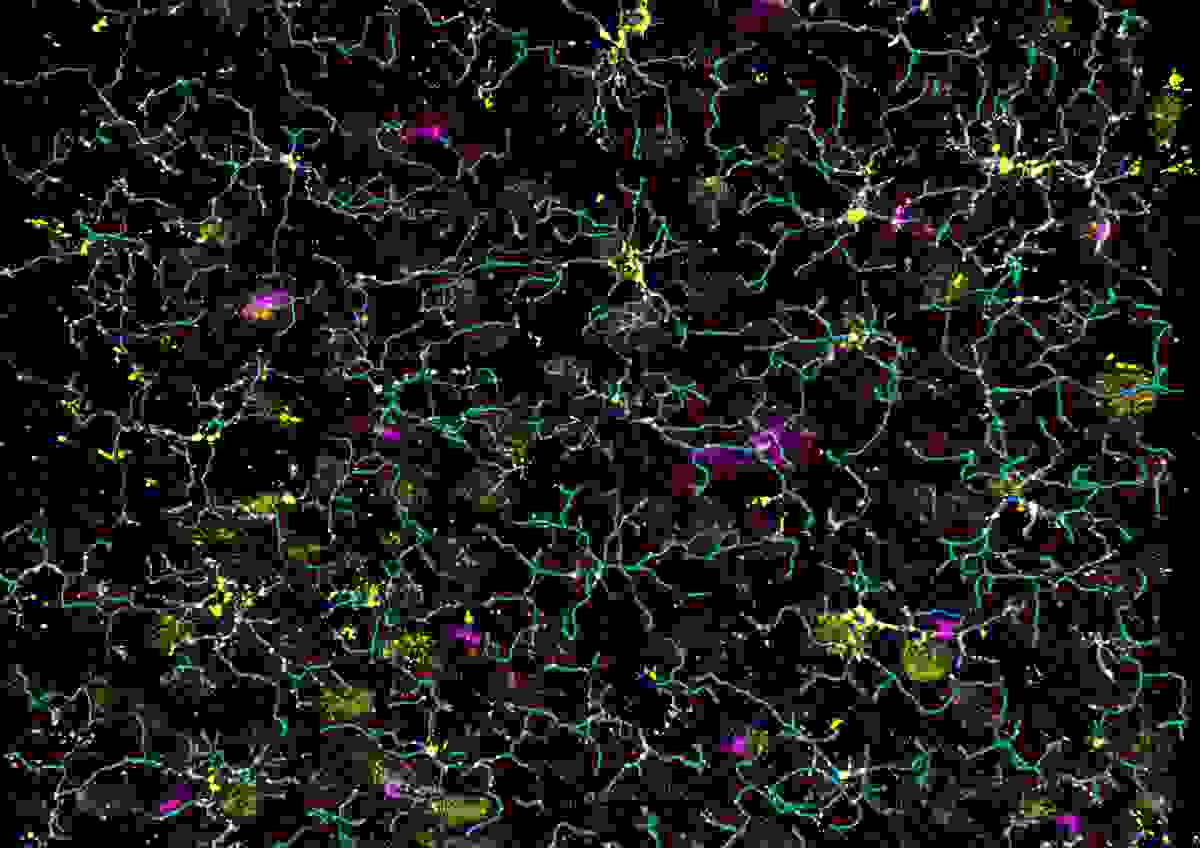
Glia
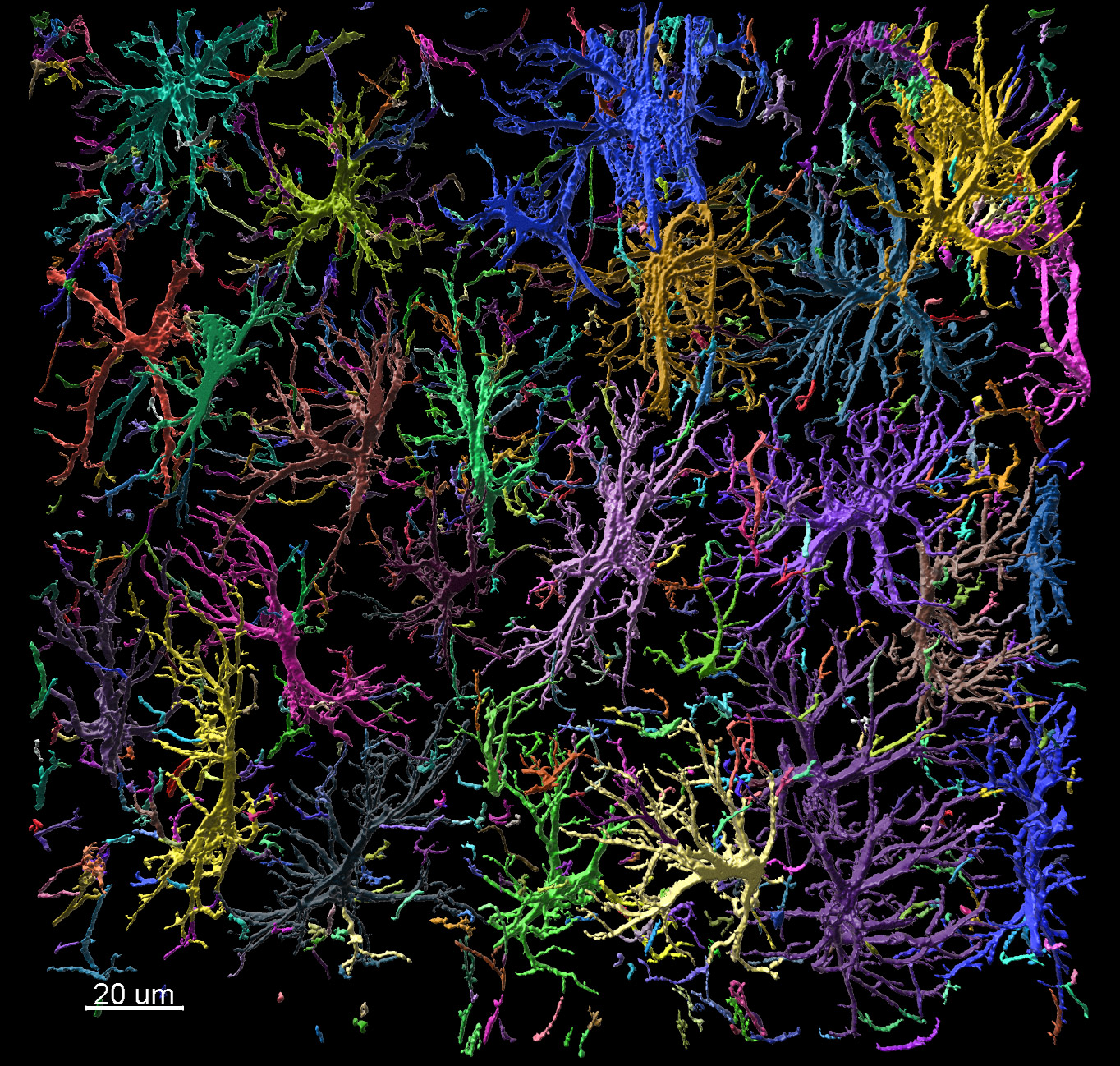
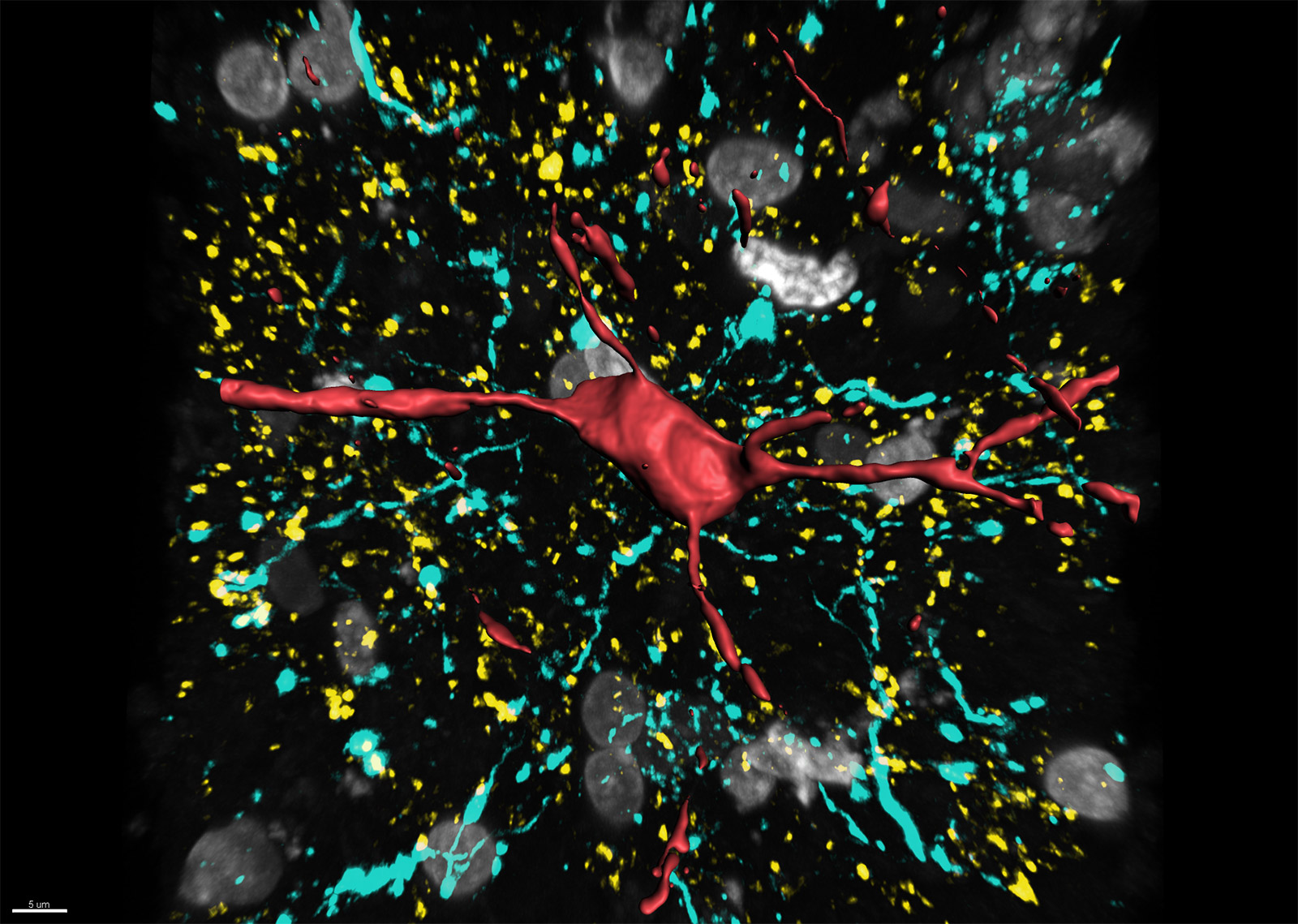
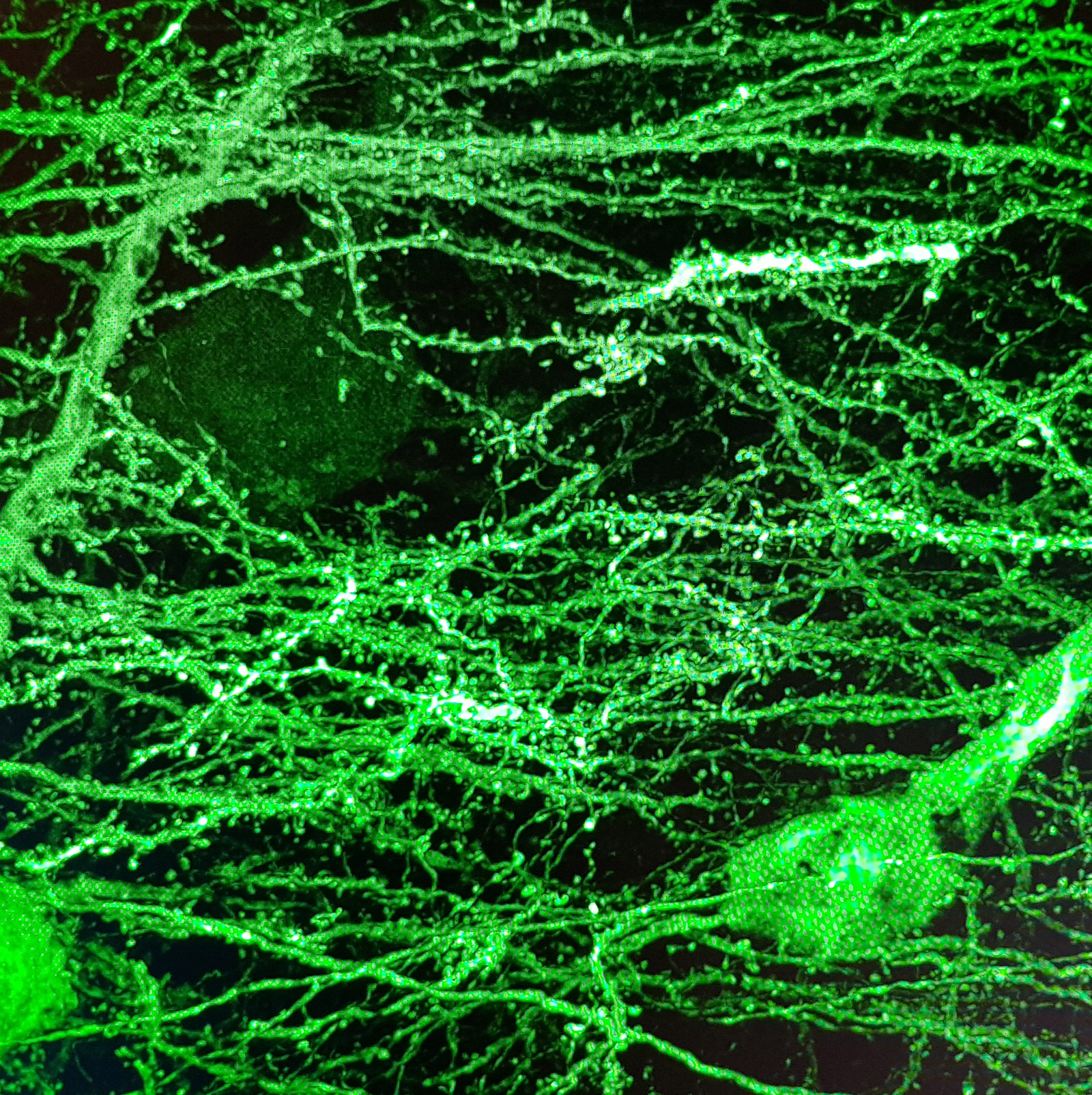
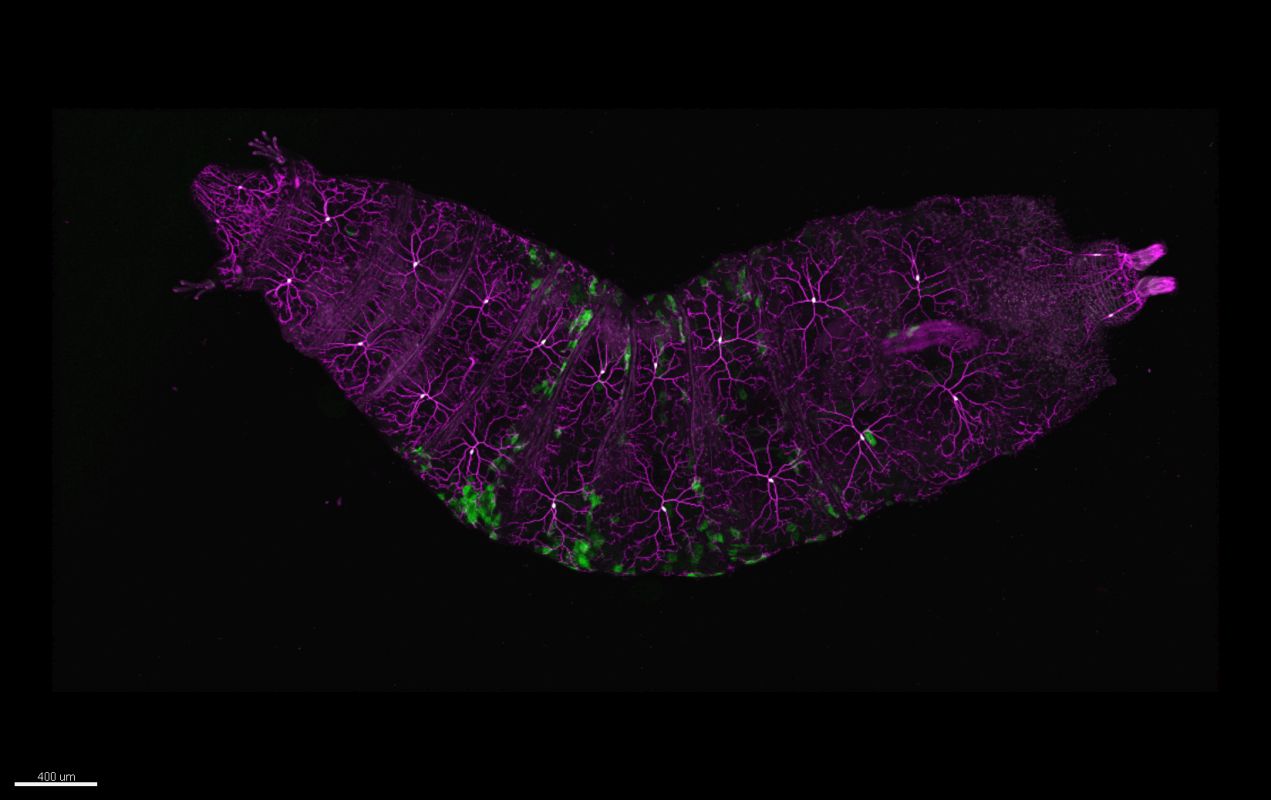
For years glial cells were presumed to have housekeeping functions that only nourished, protected and swept up after the neurons, whose role seemed more obvious. Over the last couple of decades, research into glia (consisting of microglia, astrocytes, and oligodendrocyte lineage cells) has increased dramatically, which revealed their involvement in numerous supportive but essential functions, such as supplying in nutrients and oxygen, destroying pathogens and that they even play a role in neurotransmission. Although many aspects of these cells are well characterized nowadays, the functions of the different glial populations in the brain in health and disease remain unresolved.
In vivo intravital imaging or functional calcium imaging using a fast confocal spinning disc microscope such as Dragonfly is a crucial technique to monitor glial cells and neurons under various conditions. Optogenetics devices like Mosaic can be used to manipulate glia cell functions. Number, shape and other features of microglia can be analysed with 3D image analysis software – Imaris for Neuroscientists.
Contact our Application Specialists
 Part of the Oxford Instruments Group
Part of the Oxford Instruments Group