Applications
 Part of the Oxford Instruments Group
Part of the Oxford Instruments Group
Expand
Collapse
 Part of the Oxford Instruments Group
Part of the Oxford Instruments Group
With the increasing threat of viral diseases to aquaculture, there is an urgent need for new types of vaccines that are efficient and inexpensive while also minimizing environmental risks and complying with regulations. With help from Imaris image analysis software, Rosemary Thwaite and Nerea Roher from the Universitat Autònoma de Barcelona and colleagues are developing a way to use protein nanoparticles made of fish viral antigens to create vaccines for farmed fish.
“This biomaterial provides an alternative to using soluble recombinant proteins, which cannot endure the harsh environment of the fish gastrointestinal tract,” said Thwaite. “The nanoparticles are a bioactive, robust alternative that do not require any encapsulation and can be produced in a cheap and scalable way.”
Viewing nanoparticle uptake
For the new approach, the researchers developed protein nanoparticles targeting relevant fish viruses for the aquaculture industry, including infectious pancreatic necrosis virus (IPNV), viral haemorrhagic septicaemia virus (VHSV) and viral necrosis virus (VNNV).
In addition to studying the structure of the nanoparticles, the researchers tested uptake of the nanoparticles in a zebrafish cell line. To do this they labeled the nanoparticles with Atto-488 NHS ester (green) and performed cytometry.
The researchers also imaged nanoparticle uptake with confocal microscopy to ensure the particles were inside the cell as cytometry does not distinguish between particles attached to the surface and those internalized. Using Imaris software, they analyzed the confocal images to gain a 3D view of how the particles were distributed in the cytoplasm and to visualize particles penetrating the cell membrane.
“Imaris also let us quantify the number of particles per cell and the percentage of permissive cells among those analyzed,” said Thwaite. “Being able to localize and count the particles is a useful feature of the software, and the images provided allowed us to immediately visualize what was occurring inside the cells.”
The researchers used Imaris Cell to detect cells, nuclei, and vesicles. They classified nuclei by seed points and segmented the cell using the membrane. After manually estimating the nanoparticles’ size, the researchers detected the particles as spots, allowing for different sizes, and then imported spots to vesicles, and, finally, vesicles to cell.
Quantifying particles per cell
As seen in the figure, the Imaris analysis revealed that nanoparticles targeting IPNV were taken up by 67 percent of cells with approximately 50 nanoparticles per permissive cell. Nanoparticles targeting VHSV were taken up by 100 percent of cells, averaging 88 particles per cell. These results were consistent with the cytometry data.
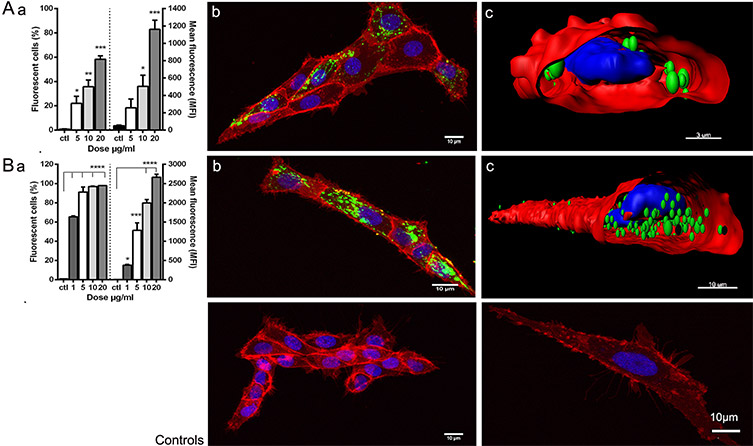
Uptake of viral antigen nanopellets (NPs) by zebrafish liver cell line (ZFL). Fluorescently labelled NPs (A) IPNV-VP2NP and (B) VHSV-G-frg16NP were added to ZFL. Control (ctl) was ZFL without NPs. (a) Cytometry: Dose-response. Cells incubated for 12 h with NPs 1-20 µg/mL in duplicate. Differences between means analysed by a one-way ANOVA with Dunnett’s multiple comparisons test, treatments versus control. Significance levels *p < 0.05; **p <0.01; ***p < 0.001; ****p <0.0001. (b) Confocal microscopy and (c) digitalized image (z-stacks) of ZFL cells after 14 h incubation with NPs at 20 µg/mL (A) and 10 µg/mL (B). NPs are green (Atto-488), cell membrane red (Cell mask Deep Red) and nuclei blue (Dapi). Control confocal images ZFL without NPs. Image courtesy of Rosemary Thwaite, Universitat Autònoma de Barcelona.
“Overall in this paper, we demonstrate the nanoparticles are taken up in vitro by zebrafish liver cells and in vivo by intubating zebrafish as a proof of concept for oral delivery,” said Thwaite. “We also showed that the nanoparticles evoke an antiviral innate immune response in vitro in gene expression studies.”
Next, the researchers plan to test the nanoparticles in farmed fish to see if the particles elicit a protective immune response against the target viruses.
The authors acknowledge N. Barba from the Servei de Microscòpia, Universitat Autònoma de Barcelona.
Author: Rosemary Thwaite, Nerea Roher and colleagues, Universitat Autònoma de Barcelona
Category: Case Study
