Applications
 Part of the Oxford Instruments Group
Part of the Oxford Instruments Group
Expand
Collapse
 Part of the Oxford Instruments Group
Part of the Oxford Instruments Group
Ophthalmology
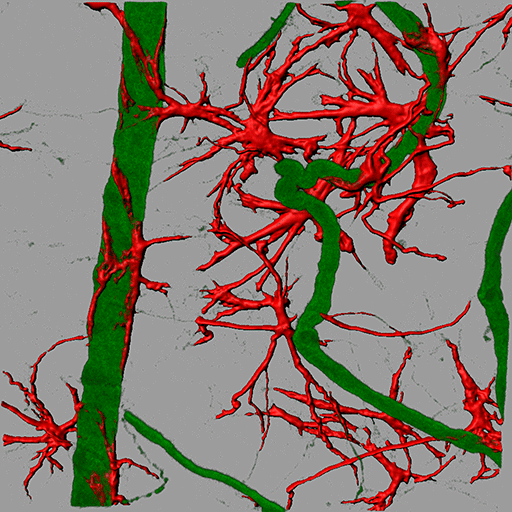
Retinal blood vessels from the retinal degeneration 1 (RD1) mouse are labeled with antibodies against the vascular basement membrane protein collagen IV (green) to decorate the vessels and glial fibrillary acidic protein (GFAP) to decorate the astrocytes (red). A tortuous (telangiectatic) vessel can be seen associated with GFAP- positive astrocytes. Surface rendering of GFAP along with blend projection in Imaris allowed the researchers to visualize these interactions. Image courtesy of Dr. Sabu Abraham.
Researchers led by Dr. John Greenwood and Dr. Stephen E. Moss from the University College London Institute of Ophthalmology are using Imaris to study vessel remodeling in the retina. Their work is revealing the mechanisms that underpin the vessel remodeling that occurs in diseases such as macular telangiectasia.
Macular telangiectasia, “wet” age-related macular degeneration, and proliferative diabetic retinopathy involve the development of new blood vessels, known as neovascularization, and/or remodeling of existing vessels. Vessel growth and remodeling is also an important feature of solid tumors and rheumatoid arthritis. Although much is known about factors that promote new blood vessel growth, less is known about vessel remodeling in disease.
To identify genes that might contribute to non-neovascular remodeling, the researchers first conducted gene expression analysis in rodent models with retinal disease that exhibited vascular pathology. Of the genes they identified, they decided to further examine the apelin gene using Imaris.
“Imaris is an excellent program for 3D rendering of 2D image stacks acquired by confocal microscopy,” says Dr. Sabu Abraham, who was part of the research team. “We found the surface rendering, filament tracing, and animation modules of the software to be very useful for analysis, interpretation and for data presentation.”
Visualizing vascular changes
The researchers used Imaris to create 3D reconstructions from confocal images of rodent retinas. From these reconstructions, they visualized the vascular changes in the presence and absence of apelin and demonstrated that apelin plays an important role in the development of telangiectatic (tortuous and dilated) vessels. The investigators also used the FilamentTracer module to quantify blood vessel parameters, which demonstrated reduced branch points in the absence of apelin. Surface rendering followed by volumetric analysis using Imaris quantified choroidal neovascular (CNV) lesion volume and revealed that there was no change in overall volume between wild type and apelin knockout mice.
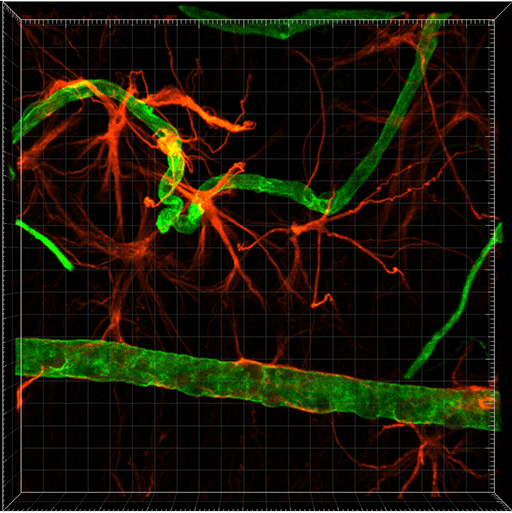
MIP Volume rendering of retinal blood vessels from the retinal degeneration mouse. Image courtesy of Dr. Sabu Abraham.
“Our studies on apelin demonstrated, for the first time, its significant role in non-neovascular remodeling in disease models and this may have implications in human disease such as macular telangiectasia,” says Dr. Abraham.
The researchers are now investigating the molecular signaling pathways associated with other novel angiogenic factors. In a recent study, they used Imaris to investigate LRG1, a newly discovered pro-angiogenic secreted protein. “Considering the immense potential of Imaris in data analysis and visualization, it will continue to be an integral part of our research methodologies,” Dr. Abraham says.

Surface rendering of glial fibrillary acidic protein (GFAP). Image courtesy of Dr. Sabu Abraham.
Research Paper: McKenzie JA, Fruttiger M, Abraham S, Lange CA, Stone J, Gandhi P, Wang X, Bainbridge J, Moss SE, Greenwood J. 2012. Apelin is required for non-neovascular remodeling in the retina. Am J Pathol. 180(1):399-409..
