Applications
 Part of the Oxford Instruments Group
Part of the Oxford Instruments Group
Expand
Collapse
Case Studies
Author: Dr. Julien Vermot and colleagues, Institut de Génétique et de Biologie Moléculaire et Cellulaire
Cell Biology
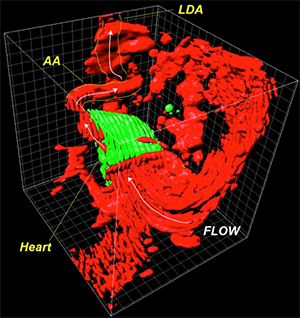
Figure 1G from paper: The researchers examined the heart and blood flow in zebrafish embryos around the time in development when blood flow began. This image shows the 3D anatomy of a heart at 26 hours post fertilization with its neighboring vasculature and flow direction. Reprinted from Goetz JG, et al., Cell Rep 6(5):799-808.
Researchers from the Institut de Génétique et de Biologie Moléculaire et Cellulaire in France used Imaris software for 3D rendering and analysis of high resolution images of live zebrafish embryo hearts around the time that blood flow first begins. Their research may lead to a better understanding of the causes of cardiovascular defects.
“The roots of the cardiovascular defects observed in adults are still unclear,” says Dr. Julien Vermot, who was part of the research team. “It is possible that some defects come from issues that begin early in life (during development) which impact later functions of the cardiovascular system.”
Formation of the cardiovascular system requires interactions among genes, fluid mechanics, and cellular reorganization. However, scientists know little about the mechanisms responsible for blood flow sensing during vascular development. The researchers wanted to understand more about the function of endothelial cilia in sensing flow conditions as the vascular network is developing.
Measuring blood flow during early development
To understand the flow conditions present when blood flow begins during early embryonic stages, the researchers acquired confocal images of live zebrafish hearts at various stages of development. They used Imaris to reconstruct 3D images, which provided 3-D data and also let them manipulate and view the beating heart in all three dimensions.
“Imaris helped us characterize the 3D motion of the different cell layers that compose the heart,” said Dr. Vermot. Specifically, they viewed various sections of a specific area of the heart’s outflow tract and characterized the cell wall motion in these sections. This analysis revealed that the embryonic heart is not efficient at pumping fluids because the inner cell layer of the heart is not opened at early embryonic stages.
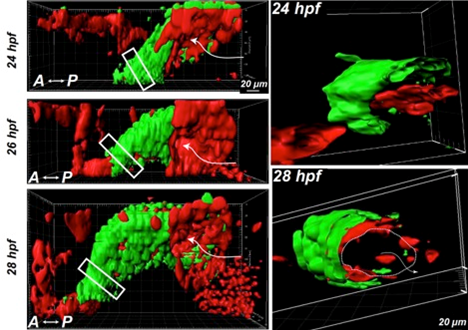
Figure 1A and C from paper: The left images show 3D imaging of the heartbeat in zebrafish embryos at 24 hours, 26 hours, and 28 hours post fertilization. The heart myocardium is labeled green (GFP), and the endocardium and blood cells are red (photoconverted Kaede). The right images show 3D sections of the boxed regions of the heart tube shown on the left. The dashed line in the section from 28 hour post fertilization shows the endocardium opening, and the arrow shows the flow direction. Reprinted from Goetz JG, et al., Cell Rep 6(5):799-808.
The researchers also conducted in vivo correlative light and electron microscopy. Overall, their analysis revealed that, at the time when blood flow begins in the embryo, endothelial cilia deflect at an angle that correlates with calcium levels in the endothelial cells. They also found that changes in shear stress, assembly of the cilia during development, or expression of the PKD2 calcium channel affected the endothelial calcium level as well as increased and perturbed vascular system development.
In future work, the researchers plan to characterize the cellular defects associated with lack of flow sensing. Dr. Vermot says that 3D analysis will be essential for this project because blood vessels are highly complex when viewed in three dimensions.