Cell Biology
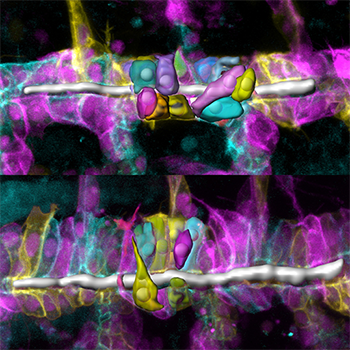
Figure 1: Single-cell analysis reveals that tracheal-cell shapes and organization are altered in Drosophila embryos with an Src42A mutation (bottom), compared to those without the mutation (top). The cell membranes are represented by the different colors.
Dominique Förster and Dr. Stefan Luschnig, University of Zurich
To function properly, organs such as the lungs, kidneys and the vascular system require tubes with specific size. However, little is known about how tube size is controlled at the cellular level. Using genetic analysis and Imaris image analysis software, Dominique Förster and Stefan Luschnig from the University of Zurich in Switzerland have discovered the cellular processes that independently regulate the expansion in length and diameter of tracheal tubes in Drosophila.
The researchers carried out image analysis on confocal Z-stacks of live Drosophila embryos. To calculate apical cell surface areas, surface elongation, and the orientation of cells relative to the tube axis, the researchers used Imaris Measurement Points to trace apical surfaces of individual cells. They then exported the X-Y-Z coordinates of single points to Matlab to project the apical outlines in the XY plane, taking into account the circumferential curvature of the cell surfaces.
To analyze cell shape, they used the Imaris Contour Drawing Mode to generate surface renderings of single tracheal cells labeled with different colors of fluorescent proteins. "(Imaris) allowed us to extract various parameters of these cell-surface objects, such as cell-surface areas and cell volumes,” Dr. Luschnig says. “We found the direct output of specific features of surface objects in the statistics menu very useful. The intuitive way of working with 3-D objects was very helpful in this context.”
They determined the longest cell axis either empirically using Measurement Points, or by extracting the ellipsoid statistics from Imaris. The coordinates of the longest ellipsoid axis (axis c) were exported to Matlab to calculate cell elongation and orientation within the tube epithelium.
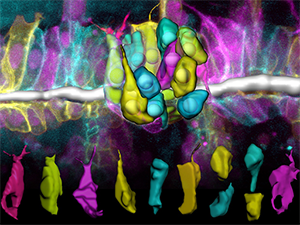
Figure 2: The cells in the Src42A mutant embryo show bias towards a perpendicular orientation relative to the tube axis.
The image analysis together with other experiments showed that the conserved tyrosine kinase Src42A acts instructively to induce polarized changes in cell shape and epithelial organization that result in tubular elongation. “Strikingly, however, Src42A controls tube elongation independently of diametric tube expansion, which is driven by apical secretion,” Dr. Luschnig says.
The results suggest that Src-dependent recycling of E-cadherin at adherens junctions is limiting for the cell shape changes that mediate tracheal tube elongation. “Our work provides a genetically tractable framework to understand the function of Src kinases in tube morphogenesis,” Dr. Luschnig says. Next, the researchers want to explore the upstream signals that regulate Src activity in a polarized fashion within the epithelium and are also trying to identify the Src substrates that mediate anisotropic cell behaviors.
Research Paper: Förster D, Luschnig S. Src42A-dependent polarized cell shape changes mediate epithelial tube elongation in Drosophila. Nat Cell Biol. 2012 Mar 25;14(5):526-34. doi: 10.1038/ncb2456.



