Applications
 Part of the Oxford Instruments Group
Part of the Oxford Instruments Group
Expand
Collapse
 Part of the Oxford Instruments Group
Part of the Oxford Instruments Group
Microbiology
Streptococcus agalactiae (Group B Streptococcus, GBS) causes serious invasive infections in newborns and adults, particularly in the elderly and individuals with underlying chronic disease. These infections are hard to fight because GBS has evolved several ways to evade the first line of immune defense. GBS can elude macrophages that would normally engulf and kill it, and if engulfed it can even survive inside a macrophage.
Although some of the bacteria’s evasive strategies are known, researchers at Perugia University in Italy wanted to find out if GBS, like some other pathogens, can manipulate the cytoskeleton of macrophages. If this was the case, understanding the mechanisms involved could lead to drug targets for treating GBS infections.
“An important part of the work was the morphological analysis of the cytoskeleton and the quantification of GBS internalized by macrophages,” said Katia Fettucciari, who led the research team. The researchers began by infecting macrophages with GBS. They labeled Vinculin as a marker of cytoskeleton alteration with anti-vinculin mouse monoclonal Ab followed by Alexa Fluor 488 goat anti-mouse IgG Ab and labeled GBS with anti-GBS polyclonal rabbit Ab followed by Alexa Fluor 568-labelled goat anti-rabbit IgG Ab. The samples were imaged with confocal microscopy and then the images were analyzed using Imaris software.
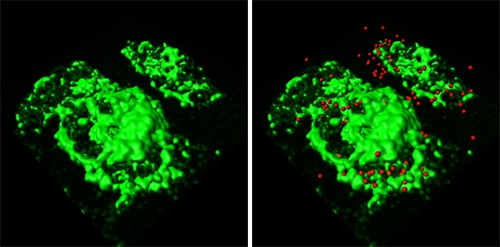
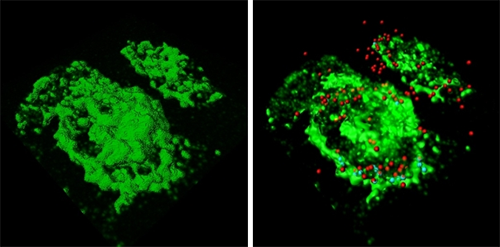
Using confocal images, the researchers used the Imaris iso-surface function to reconstruct GBS-infected macrophages in 3D (figure 1a). They pinpointed the location of GBS with red spot objectss using the Surpass module (figure 1b) and then used the Cell module to define macrophage volume (figure 1c). They also identified and enumerate internalized (figure 1d, cyan) and external GBS (figure 1d, red).
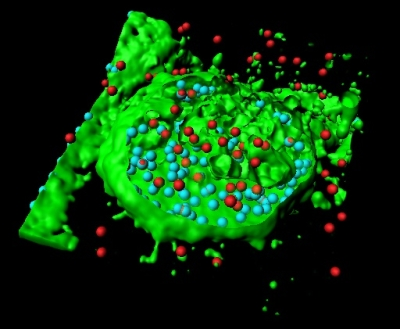
Figure 2: Average cell velocity. Circles represent average cell velocities of individual cells, white bars indicate median values.
The Imaris Cell module, part of Imaris suite - the most powerful and versatile 3D and 4D image analysis software solution on the market for researchers in life sciences, enabled a precise and reproducible quantification of particles inside and outside a chosen area or volume, said Fettucciari. The researchers reconstructed macrophages in 3D with the software’s iso-surface function (Figure 1a) and pinpointed the locations of external GBS with red-colored spot objects using the Imaris MeasurementPro (Figure 1b). Then they used the Cell and Imaris XT modules to precisely define the macrophage volume (Figure 1c, green) and to identify and enumerate internalized (figure 1d, cyan) as well as external GBS (red). The statistical functions of Imaris calculated the ratio between intracellular and extracellular GBS. When the researchers performed the same analysis on images of samples treated with a calpain inhibitor, they found that the intracellular GBS increased dramatically (Figure 2). Inhibiting calpain led to more phagocytosis because GBS cytoskeletal degradation was hindered.
The image analysis together with cell assays showed that GBS can evade macrophage antimicrobicidal defenses by cleaving several structural and regulatory cytoskeletal proteins, which leads to cytoskeleton disruption. The mechanism for this disruption is unusual for bacteria and is mediated by calcium influx and calpain activation. GBS disrupts the dynamics of the macrophage actin and microtubule cytoskeleton, which are necessary for its uptake. Because GBS avoids macrophage phagocytosis and subsequently intramacrophage antimicrobial responses the infection spreads as the bacteria replicate extracellularly. Fettucciari said that these findings could be important for the management of GBS infection because they point to inhibition of calpain activation as a key target.
Research Paper: Group B Streptococcus (GBS) disrupts by calpain activation the actin and microtubule cytoskeleton of macrophages, Cellular Microbiology, DOI: 10.1111/j.1462-5822.2011.01584.x
