Applications
 Part of the Oxford Instruments Group
Part of the Oxford Instruments Group
Expand
Collapse
 Part of the Oxford Instruments Group
Part of the Oxford Instruments Group
Microvascular Research
Researchers led by Dr. Joel S. Pachter at the University of Connecticut Health Center are studying tight junction proteins, which are a critical element of the blood-brain barrier. By incorporating Imaris into a new 3D analysis approach, they can study where and how tight junction proteins are regulated.
The blood-brain barrier limits the passage of molecules and cells from peripheral circulation into the central nervous system. Understanding how this barrier operates could help scientists to enhance the delivery of drugs across the barrier and provide a better understanding of how it breaks down during diseases such as the neuroinflammatory condition multiple sclerosis.
Dr. Pachter’s research team is focused on the Claudin-5 (CLN-5) tight junction protein. “The use of Imaris to perform isosurface rendering of junctional CLN-5 staining and 3D contour-based segmentation of spinal cord microvessels enabled us to isolate specific microvessels in a 3D confocal dataset, which allowed us to assess the CLN-5 density within their endothelium,” says Debayon Paul, who is a member of the research team. “This gave us a measure of which microvessels were being damaged during neuroinflammation, and to what degree.”
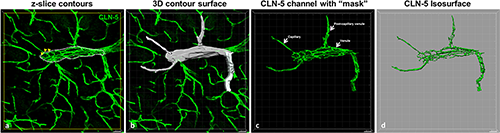
Figure 1. 3D contour-based quantification of junctional CLN-5 in spinal cord microvessels. CNS microvascular tributaries (e.g. capillaries, post-capillary venules) emerging from a venule of a naïve mouse detailing CLN-5 (green) staining at intercellular junctions. (a) To quantify CLN-5 staining associated with a microvessel in 3D, an individual contour for each confocal z-slice was created by cursoring out the vessel of interest (yellow arrows). (b) The individual z-slice contours were merged into a 3D contour surface. (c) This contour surface was utilized to isolate the microvessel of interest by masking, and its area was used as an estimate of the microvascular surface area. (d) An isosurface for the CLN-5 channel was created from the selected vessel for statistical analysis.
The researchers acquired confocal images of 60-micron thick spinal cord cryosections from mouse tissue and then used Imaris to visualize and quantify CLN-5 density in small-diameter capillaries with closely apposed or connected venular structures. These small capillaries are challenging to analyze using thin sections. They used Imaris for microvascular segmentation by generating a 3D contour surface. This approach allowed more accurate approximation of the 3D surface area of the microvascular endothelium. The investigators calculated the CLN-5 density from the number of CLN-5 voxels and their mean intensity in the enclosed contour surface.
The study also made use of the Clipping Plane tool, which truncates the image along a given plane in 3D space by cutting away objects on one side of the plane. “The Clipping Plane tool on Imaris helped in resolving the interior versus exterior (luminal versus abluminal) vascular compartments, and the microvascular basement membrane into its endothelial and parenchymal counterparts, which split apart during neuroinflammation to accommodate the CNS invading leukocytes,” says Dr. Pachter.
Their 3D analysis revealed heterogeneity in CLN-5 protein expression, during normal and pathological states, in central nervous system microvessels, which include venules, post-capillary venules and capillaries. Under normal conditions, the density of CLN-5 staining was the greatest in capillaries and smaller venules, and the staining was the least in the larger venules. However, in mice inflicted with experimental autoimmune encephalomyelitis, a model for multiple sclerosis, this heterogeneity was exacerbated.
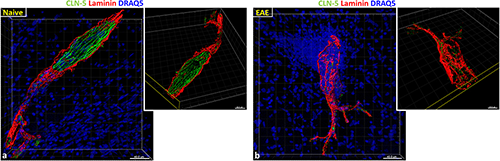
Figure 2. CLN-5 staining in spinal cord microvessels from naïve vs. EAE mice. Isosurface rendered CLN-5 (green) and microvascular basement membrane protein Laminin (red) staining in confocal z-stacks. Nuclei are highlighted with DRAQ5 (blue). (a) Naïve venule. Inset showing intact TJ staining and view through lumen cut using clipping plane. (b) Inflamed venule showing the close association of altered junctional CLN-5 with dense perivascular cellularity (reflective of leukocyte infiltrates) during EAE. Inset highlights the fragmented loss of CLN-5 staining and damaged basement membrane.
“By subjecting immunostained microvessels in thick sections to 3D rendering with Imaris, we could obtain more accurate focal information and an enhanced spatial perspective regarding tight junction protein staining in naïve and inflamed microvessels,” Dr. Pachter adds. “This yielded both qualitative and quantitative information about heterogeneity in the behavior of different-sized CNS microvessels under normal and neuroinflammatory settings.”
The 3D analysis method should be readily adaptable for analysis of other junctional proteins of the CNS and peripheral microvasculature. “Having established the idea about where (which microvessels) leukocytes preferentially egress from the circulation during neuroinflammation, we next aim to apply this 3D visualization and quantification approach to address how leukocytes cross the endothelial and astrocytic barriers that comprise the BBB, to enter CNS parenchyma,”Dr. Pachter says.
Research Paper: Paul D, Cowan AE, Ge S, Pachter JS. Novel 3D analysis of Claudin-5 reveals significant endothelial heterogeneity among CNS microvessels. Microvasc Res. 2013 Mar;86:1-10. doi: 10.1016/j.mvr.2012.12.001..
Author: Debayon Paul, Dr. Joel S. Pachter and colleagues, University of Connecticut Health Center
Category: Case Study
