Applications
 Part of the Oxford Instruments Group
Part of the Oxford Instruments Group
Expand
Collapse
 Part of the Oxford Instruments Group
Part of the Oxford Instruments Group
Plant Biology
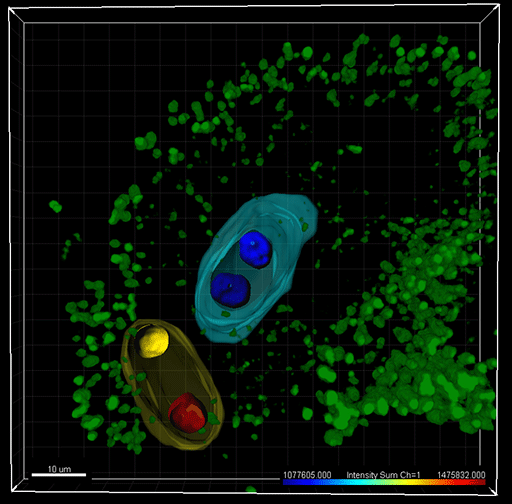
The RNA helicase MNEME (MEM) restricts germline fate in plant ovules. The image shows a mem mutant ovule in Arabidopsis: an ectopic gametophyte (yellow) containing unreduced nuclei has developed alongside a normal embryo sac (blue) containing reduced, haploid nuclei. Imaris was used for 3-dimensional image reconstruction, in silico nuclei isolation and statistic reporting of fluorescence intensity. The nuclei were counterstained with a GFP-chromatin marker and nuclei surfaces are color-coded according to the intensity sum. Surfaces of the gametophytes are pseudo-colored (yellow, blue). Green: chloroplasts autofluorescence in the ovule. Image courtesy of Célia Baroux.
A key step of sexual plant reproduction is sporogenesis, which mediates the transition from the sporophyte to the generation of a gametophyte. Drs. Baroux, Schmidt, and Grossniklaus from the University of Zurich used Imaris image analysis software to help them understand the mechanisms involved in sporogenesis, revealing information that could help in engineering crops with desirable genotypes.
In plants, the first cell of the female germline, called the megaspore mother cell (MMC), undergoes meiosis to form a tetrad of reduced megaspores. The female gametophyte, which contains the female gametes, forms through mitotic divisions from one of these megaspores. After double fertilization of the two gametes, they form the embryo and the endosperm found inside the developing seed.
Quantitative analyses
Drs. Baroux, Schmidt, and Grossniklaus used Imaris to perform quantitative analyses of cell identity markers at the single-cell level and in whole-mount plant tissues. They conducted 3-dimensional image reconstruction in Surpass mode, in silico object isolation using contour surfaces, and fluorescent signal quantification using the software’s statistical tools.
“Imaris allows us an unprecedented qualitative and quantitative resolution in understanding cell fate determination and cellular organization during plant reproduction,” Dr. Baroux says. “Imaris is an essential tool in our research.”
The researchers first analyzed cell-type specific transcriptional profiles. They identified the RNA helicase MNEME (MEM) as predominantly expressed in the MMC and discovered that MEM restricts germline fate to one cell per ovule. More than one MMC-like cell forms in mem mutant plants, leading to the development of additional female gametophytes. The researchers used Imaris to determine the ploidy level of the additional gametophyte(s) in mem mutants.
The investigators used H2B-YFP as a fluorescent marker under the control of the gametophytic AKV-promoter. They took a series of confocal stacks of mem mutant and wild-type cells and then reconstructed images in 3D mode. They used the Imaris surface tool to draw contour surfaces around each nucleus, which expressed YFP. They also quantified the fluorescence intensities in the fluorescence channel by obtaining values for the sum of voxel intensities in the 3D-reconstructed images.
Precise measurements
“Imaris is highly efficient for the precise measurement of intensity values for the selected 3D volume elements,” Dr. Schmidt says. “Its capability to measure fluorescence intensity for a selected region of 3D objects, combined with its speed and precision, was most useful for this study.”
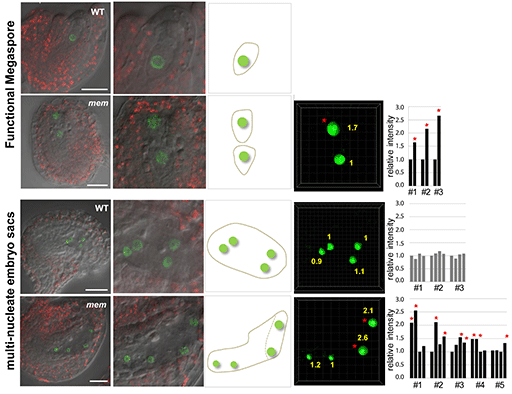
The researchers used Imaris to quantify fluorescence intensity in ovules with single and double gametophytes to estimate ploidy levels. The figure shows the relative YFP fluorescence intensity as quantified in gametophytic nuclei of mem-1 and mem-2 heterozygous mutant plants harboring either one or two developing gametophytes expressing the H2B-YFP marker under the control of the AKV-promoter. The wild-type (WT) picture shows an ovule with wild-type morphology from a mem-1/MEM plant; other pictures are from mem-2/MEM mutants; scale bars 20 microns. In ovules harboring two distinct gametophytes at early stages of female gametophyte development, the relative signal intensity from the first gametophyte (developing at the normal position) and the second, additional gametophyte (usually developing in a more micropylar region) differ: a similar intensity as in WT was observed in the first gametophyte, while a higher intensity was observed in the second gametophyte (labeled by *). While in WT the ploidy level of gametophytic nuclei is haploid, the higher signal intensity in additional gametophytes indicates a higher ploidy level, suggesting that these additional gametophytes developed from a somatic cell without meiotic reduction. Importantly, this effect was observed in ovules harboring two post-meiotic, one-nucleate female gametophytes and in ovules with four gametophytic nuclei (in mem mutants likely belonging to two female gametophytes), making it unlikely that the increase in fluorescence level results from alterations during the first mitotic division of the gametophyte. Image courtesy of the authors.
The Imaris analysis revealed that the gametophytes in the mem mutants had a higher ploidy level than the wild-type cells, indicating that they were formed from a somatic cell, i.e. through an apomictic process. The fluorescent signal intensities were approximately two times higher in the nuclei of the additional gametophytes than in the wild type, demonstrating their unreduced nature.
The investigators continue to use Imaris as they move forward in this research. “We are extensively using Imaris to quantify cellular and chromatin markers in whole-mount reproductive tissues, such as in developing ovules and pollen of Arabidopsis plants,” Dr. Baroux says. “For this, we are also using Imaris for 4D reconstruction and for deriving 4D statistics in object tracking.”
Research paper: Schmidt A, Wuest SE, Vijverberg K, Baroux C, Kleen D, et al. (2011) Transcriptome Analysis of the Arabidopsis Megaspore Mother Cell Uncovers the Importance of RNA Helicases for Plant Germline Development. PLoS Biol 9(9): e1001155. doi:10.1371/journal.pbio.1001155
