Applications
 Part of the Oxford Instruments Group
Part of the Oxford Instruments Group
Expand
Collapse
 Part of the Oxford Instruments Group
Part of the Oxford Instruments Group
Mario R. Capecchi and colleagues from the University of Utah School of Medicine in Salt Lake City are using Imaris to study the mechanisms that drive neuropsychiatric diseases such as obsessive-compulsive spectrum disorders, or OCD. More in-depth knowledge of the disease’s mechanisms could lead to new treatment options and medications.
Examining brain immune cells
The researchers previously developed Hoxb8 knockout mice mouse models that exhibit compulsive behavior similar to OCD. Observations in these mice strongly suggested that the neuroimmune system, specifically microglial cells, are important in the disease pathology. To find out more, the researchers used Imaris to study a subset of microglia, called the Hoxb8 microglia, that have a regional- and sex-dependent distribution in the mouse brain. They explored how these microglia behaved as well as their developmental origin.
“This work is part of a careful dissection of the cellular and molecular pathological events to identify effective therapeutic targets that would provide a better quality of life for patients with OCD,” said Capecchi. “Imaris allowed us to precisely and reliably analyze all the different facets of microglia behavior, including movement, distribution and density, morphology, and phagocytosis, with one software package.”
Microglial cells are part of the brain’s immune system and play an important role by removing plaques, damaged or unnecessary neurons and synapses, and infectious agents. These dynamic cells feature a highly branched morphology that includes processes that constantly move and are very reactive to changes in their environment.
Live brain imaging
The researchers used multiphoton microscopy to image the brain of living mice through an open skull window. They imaged microglial cells 50-80 microns below brain surface at a resolution of 1024×1024 pixels, acquiring images at 0.05 frames per second for 30-35 min.
The researchers used Imaris to analyze the multiphoton images as well as confocal images they acquired to better understand how Hoxb8 microglia enter the embryonic brain, develop over time, and their distribution with respect to key brain regions associated with OCD. Imaris also helped the researchers observe the role of these microglia in the development and maintenance of the neural tissue.
Specifically, they used the Imaris Spots function to count the number of microglia per unit area and then identified subsets of cells that were co-labelled with markers for proliferating cells, apoptotic cells, etc. by filtering the mean intensities of the marker’s fluorescence. They also quantified the area of the analysis region using the Imaris Surface function.
To study how the Hoxb8 microglia respond to injury in the brain, the researchers created a focused laser injury to a small area within the region being studied. They assessed how fast the microglial processes moved towards the injury by first identifying the site of injury as a point of reference with Surfaces. They then performed the Imaris Xtension functions ‘time subtract average’ to exclude the stationary site of injury from movement/tracking calculations and ‘drift correction’ to correct artifacts caused by the movement of the animal during imaging.
They also followed the motion of the microglial processes by identifying the bulbous ends of the processes with Spots and then using the Imaris TrackLineage ‘Autoregressive motion’ algorithm to identify the movements, or tracks, of these processes through the entire time period. Finally, running the Xtension ‘Distance transformation’ on the site of injury surface produced numerical values for parameters such as speed and distance that were used to analyze movement of the microglial processes.
Effects of microglia dysfunction
Data from image analysis and additional experiments suggests that Hoxb8 microglia dysfunction in specific brain regions impacts neural circuitry in a way that manifests as behavioral defects, such as obsessive grooming, high anxiety, and social impairment.
The researchers plan to take this work further by genetically and pharmacologically manipulating different subsets of microglia and then assessing the impact on animal behavior. These experiments will help identify specific conditions that rectify microglial dysfunction in the OCD disease model and reduce OCD-like behavior. They plan to continue to use Imaris to analyze the changes in microglia behavior that result from various methods of manipulation.
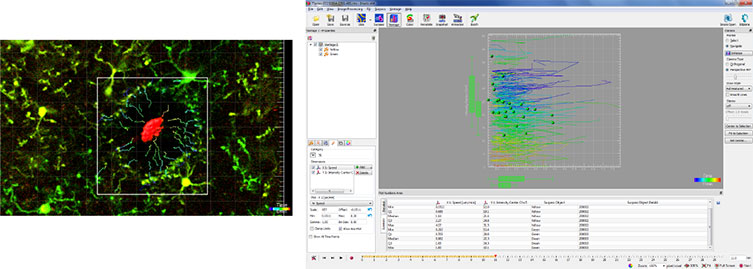
These images demonstrate the process of calculating filopodia movement of microglia in response to a focused laser ablation in the brain of a live anesthetized mouse. To assess the speed of the microglial processes moving towards the site of focused laser injury, the site of injury was identified with Imaris Surfaces to set it as the point of reference. The researchers then performed the Imaris Xtension function Time Subtract Average to exclude the stationary site of injury from the subsequent movement/tracking calculations and performed drift correction to correct for any artifacts caused by the movement of the test subject during image acquisition. Next, the microglial processes, were identified with Spots and the TrackLineage Autoregressive motion algorithm was used to identify the movements (tracks) of these processes through the entire time period. Finally, running the distance transformation Xtension on the site of injury surface produced the numerical values of the different parameters, such as speed and distance, that were used to analyze the movement of the microglial processes. Courtesy of Mario R. Capecchi, University of Utah School of Medicine.
Author: Mario R. Capecchi and colleagues, University of Utah School of Medicine in Salt Lake City
Category: Case Study
