Applications
 Part of the Oxford Instruments Group
Part of the Oxford Instruments Group
Expand
Collapse
 Part of the Oxford Instruments Group
Part of the Oxford Instruments Group
Light Sheet Microscopy
Thomas Liebmann and colleagues are using an imaging method called immunolabeling-enabled 3D imaging of solvent cleared organs (iDISCO) to better understand the pathological hallmarks of Alzheimer’s disease. They used the technique — which combines immunolabeling, light sheet microscopy, image analysis with Imaris, and tissue clearing — to produce 3D renderings of an entire brain hemisphere of a mouse model of Alzheimer’s disease as well as for volume imaging of human frozen tissue samples.
The analysis revealed important differences between the pathology of Alzheimer’s disease in the brains of animal models and in the brains of people with the disease. This new information could eventually lead to a better understanding of the initial causes of Alzheimer’s disease.
The unknowns of Alzheimer’s
Pathologically, Alzheimer’s disease is characterized by 1) an abnormal accumulation of the beta amyloid peptide, which aggregates and leads to amyloid plaques in the brain and 2) the presence of intracellular tau protein inclusions known as neurofibrillary tangles. Although Alzheimer’s pathology has been intensively studied, scientists still aren’t sure about the origin and the role of the amyloid plaques and neurofibrillary tangles. Liebmann and his colleagues want to better understand the three dimensionality of these disease hallmarks and their complex association with the immediate environment, which could help reveal underlying mechanisms of neurodegeneration.
“The approach we demonstrated allows researchers and clinicians to detect and characterize the progression of Alzheimer’s disease symptoms with an entirely new perspective,” said Liebmann. “As fundamentals of the disease causation are still debated, the addition of the large-volume context could lead to new insight.”
Using iDISCO, the researchers labeled and performed volume imaging of amyloid, tau protein, cell nuclei, microglia, and vasculature. With this method, they could visualize up to three different labels simultaneously.
The researchers used Imaris for visualization in 3D, anatomical segmentation of the tissue, intensity quantification, spatial distribution measurements, and animated videos for dynamic and comprehensive representation of the imaging data. “We used Imaris because of its ability to render and segment surfaces in large volumes for many image channels simultaneously,” said Liebmann.
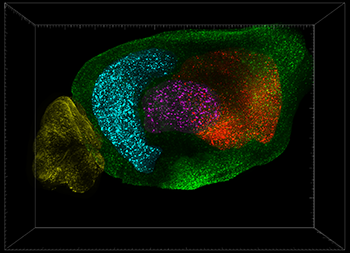
This image from a 2-year-old mouse model of Alzheimer’s disease is stained for beta amyloid. Plaques in different major regions of the brain are pseudo colored. The regions are the cortex (green), hippocampus (cyan), striatum (red), cerebellum (yellow), thalamus (magenta). Image courtesy of Thomas Liebmann, The Rockefeller University.
The researchers used Imaris to digitally dissect imaging volumes by manually tracing autofluorescence channels at appropriately spaced planes. They also conducted automated surface rendering for small volumes of amyloid plaques, glia, and vessels using Imaris.
To study the progression of amyloid plaque accumulation in a mouse model of Alzheimer’s disease, the researchers used Imaris spot detection to automatically detect amyloid plaques in each digitally dissected region of the mouse brain. This analysis revealed that the amyloid plaques started in the cortex and, surprisingly, increased in the cerebellum earlier than in the hippocampus in one particular Alzheimer’s disease mouse model.
“Light-sheet microscopy allowed us to efficiently image the entire mouse brain without the need for mechanical sectioning and laborious image reconstruction,” said Liebmann. “All volume representations from the light-sheet microscope were rendered using the Imaris Surpass mode, and videos were rendered using the key frame animation mode.”
They also applied the iDISCO technique to volume analysis of archived human brain tissue, which revealed clear differences from the mouse brains. “We found strikingly unique plaque patterns in the human Alzheimer’s brain, which we call TAPs, or three dimensional amyloid patterns,” said Liebmann. “This study revealed limitations in the use of animal models for studying the disease and suggests a need for detailed retrospective studies to correlate TAP patterns with clinical evaluation. Ultimately, these correlations could help us target the disease with a more personalized approach.”
The researchers add that this imaging approach opens up new types of quantitative preclinical studies and could be used for pathological exploration through correlative examination using retrospective studies of archival tissues.
