Applications
 Part of the Oxford Instruments Group
Part of the Oxford Instruments Group
Expand
Collapse
 Part of the Oxford Instruments Group
Part of the Oxford Instruments Group
Cell Biology
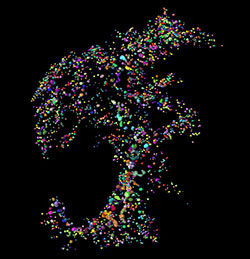
This Imaris isosurface rendering show an OPT-imaged pancreas from a healthy C57BL/6 mouse (splenic lobe). The insulin-producing islets of Langerhans are pseudocolored to illustrate the ability of Imaris to cross-reference volumetric and spatial data. Each color corresponds to an islet with known volume and spatial x, y, z coordinates. The specimen measures approximately 1 by 1.4 centimeters.
Researchers led by Ulf Ahlgren, of Umeå University, in Sweden, are using Imaris software with optical projection tomography (OPT) to compare beta cell distribution in the pancreas of a mouse model of diabetes with that of normal mice. The new work not only reveals never-before-seen details of pancreas pathophysiology in these mice, but also provides a new comprehensive record of how these insulin-producing cells are distributed in the murine pancreas.
3D imaging of beta cell distribution
The pancreas is a mixed endocrine and exocrine gland that plays a pivotal role in maintaining blood glucose homeostasis. The endocrine component, organized into the islets of Langerhans, constitutes only 1 to 2 percent of the total pancreatic mass and is scattered throughout the volume of the gland. Insulin-producing beta cells are the most prominent cell type in the islets of Langerhans.
The researchers were interested in developing a better picture of beta cell distribution in the pancreas to fully understanding the pathophysiology of animal models of diabetes and metabolic stress. Although beta cell distribution has been studied using stereological approaches, these methods rely on the extrapolation of two-dimensional data. They are, therefore, limited for examining changes in the number of beta-cells in the pancreas, particularly if the changes aren’t the same throughout the organ.
The investigators had previously developed a range of OPT protocols that can extract quantitative data and spatial coordinates of individual molecularly labelled objects throughout the volume of the intact rodent pancreas. Combining their OPT protocols with analysis with Imaris allowed the researchers to study beta cell distribution in the leptin-deficient (ob/ob) mouse on the whole-organ scale. This mouse model, which exhibits obesity and insulin resistance, is widely used to study metabolic disturbances that lead to type 2 diabetes.
“The ability of Imaris to extract combined volumetric and 3D-spatial information from our OPT data has been an invaluable asset to us in a number of studies,” said Ahlgren. “In particular, the possibility to cross-reference the volumetric and spatial data has allowed us to perform detailed assessments of how islets of certain size categories are affected in the model systems we investigated.”
Analyzing optical projection tomography data
After acquiring OPT images of mice pancreata, the researchers quantified beta cell volumes by using Imaris to create 3D isosurfaces of the cells. This revealed that internal islet lesions commonly form in the leptin-deficient mice. The researchers then created 3-D isosurfaces of the lesions to quantify their volumes.
“Despite thousands of previous papers describing this mouse model, these lesions have been overlooked,” said Ahlgren. “Using 3D isosurfaces created with Imaris, we demonstrated that these lesions could constitute as much as 15 percent of the overall islet volume in the pancreas.”
Once they had created the isosurfaces, the researchers used Imaris to pseudocolor islets based on their volume and spatial x, y, z coordinates and to pseudocolor islet beta cells according to small, medium, and large volume size categories. This allowed observations of how certain categories of islets, relating to their size or spatial distribution, are affected by diabetes disease progression in the pancreas. This type of analysis would be extremely challenging using existing methodologies.
Based on their findings, the researchers hypothesize that in the leptin-deficient mice, a hyperglycemic state increases demand for insulin, which leads to increased blood flow, vessel dilation and altered vascular structure. This, in turn, causes red blood cells/plasma to leak from vessels in the islets. The extravasated red blood cells then clot and form a fibrin mesh, creating lesions within the islet core that may affect islet function (Scientific Reports 2016).
Comprehensive data sets
The researchers detail their tomographic data and Imaris files in a related paper (Scientific Data 2017). The published data sets include the full distribution of beta-cell mass throughout the volume of the pancreas in groups of obese ob/ob mice and their lean controls between 4 and 52 weeks of age. Also included is information about individual islet beta cell volumes, including their three-dimensional coordinates, and shape.
“These new data sets comprise the most comprehensive public record of the beta-cell mass distribution in the mouse,” said Ahlgren. “They may serve as a powerful reference for planning a variety of in vivo or ex vivo experiments, including computational modelling and statistical analyses, that involve the rodent pancreas.”
According to the researchers, the data sets also provide a powerful tool for planning stereological sampling assessments by shedding light on intra- and intevideor-lobular variations in beta cell distribution.
